The award is part of the i3 Center’s strategy to tighten collaborative cross-disciplinary interactions between cancer immunologists and materials scientists to advance immunotherapy
By Benjamin Boettner
(BOSTON) — The Harvard “Immuno-engineering to Improve Immunotherapy” (i3) Center at the Wyss Institute, which is funded by the National Institutes of Health (NIH) as part of its Cancer Moonshot initiative to advance innovative biomaterials-based anti-cancer immunotherapies, announced that its first grant award to physician-scientist Eric Smith, M.D., Ph.D. Smith is Director of Translational Research for Immune Effector Cell Therapies at Dana-Farber Cancer Institute (DFCI).

He will address current challenges of extending CAR-T cell therapies to the treatment of solid tumors by deploying immuno-materials-based approaches developed by Wyss Founding Core Faculty member David Mooney, Ph.D., and his team at the Wyss Institute and the Harvard John E. Paulson School of Engineering and Applied Sciences (SEAS). The award to Smith is the first of several awards that will be annually given to researchers to bring additional expertise and perspective to the Center and strengthen its collaborative multi-disciplinary approach towards solving key challenges in immunotherapy.
For CAR-T cell therapies (chimeric antigen receptor-T cell therapies), T cells are collected from patients and genetically engineered outside of the body to express an artificial T cell receptor molecule on their surface. Once CAR-T cells are administered to patients, the added receptor allows them to specifically recognize cancer cells and unleash a potent T cell response that destroys them. While CAR-T cell therapies have successfully eliminated blood cancers in patients, with first therapies approved by the Federal Drug Administration (FDA) to treat certain types of recurring leukemias and lymphomas, they have not yet shown similar efficacies against solid tumors.
“I am honored to be awarded this grant which will combine nanomaterials with next-generation adoptive cellular therapy approaches in an attempt to address the challenge of extending this therapeutic modality that has been so transformative for patients with difficult-to-treat hematologic malignancies to solid tumors,” said Smith, who also is Principal Investigator of a synthetic biology and cellular engineering pre-clinical discovery laboratory group.
His early work identified specific cell surface targets in multiple myeloma tumors with limited predicted “on target-off tumor” potential, including the novel target for myeloma immunotherapy known as GPRC5D. He engineered, evaluated, and optimized fully human CAR vectors targeting both GPRC5D and BCMA, another cell surface protein expressed by myeloma cells, and there are currently six clinical trials based on this work. Ongoing efforts in his lab focus on understanding sub-optimal responses of cellular therapies and, through using the latest genetic engineering and cell manufacturing tools, designing solutions around these pitfalls.
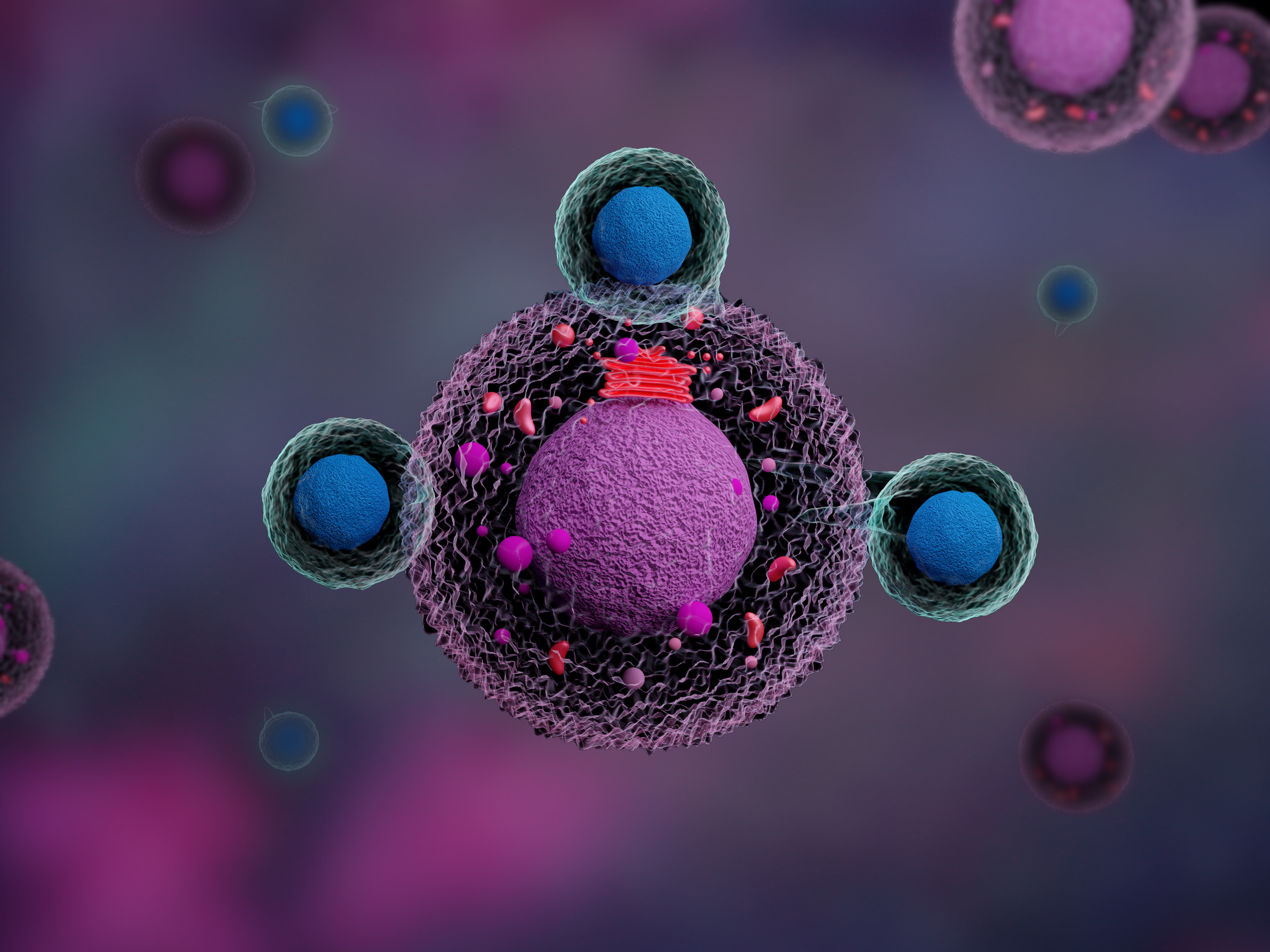
Smith is working on new solutions to challenges of extending CAR-T cell therapies to the treatment of solid tumors. Shown is a microscopic image of a CAR-T cell as it attacks tumor cells in its vicinity. Credit: Shutterstock/ Meletios Verras[/caption] Cancer immunologists believe that a large part the ineffectiveness of CAR-T cell therapy in solid tumors is due to the difficulty of the provided CAR-T cells to accumulate inside or close to the microenvironment of a solid tumor in sufficient numbers, as well as the ability of tumor cells to restrain resident and incoming immunogenic T cells, or even switch immune cells into an inhibitory state in which they actively suppress anti-tumor immunity. To enable CAR T cell expansion, oncologists need to apply chemotherapies that generally deplete immune cells and eliminate potentially inhibitory immune cells before CAR-T cells are given to patients (lymphodepleting conditioning chemotherapy). This creates room for CAR-T cells to expand and gives them an advantage over inhibitory T cells that they have to compete with. Very frequently, however, lymphodepleting conditioning chemotherapy is not effective enough, and it temporarily paralyzes the immune system so that patients become vulnerable to infection with pathogens and other complications.
Smith and his group at the DFCI will use the award to explore immuno-materials called antigen-presenting cell (APC) mimetic scaffolds, which have been shown by Mooney’s team to efficiently expand CAR-T cells ex vivo, for their potential to also expand CAR-T cells when placed into or close to a solid tumor inside the body. The researchers hope that this could open a path to significantly enhance the numbers of active CAR-T cells inside tumors and obviate lymphodepleting conditioning chemotherapy.
In a second independent aim Smith will focus on combining CAR-T cell therapy with checkpoint inhibitor therapy, another form of immunotherapy that by now has been proven to successfully help shrink or eliminate a range of solid tumor types, and in 2018 garnered the Nobel Prize in Physiology or Medicine. Checkpoint inhibitor drugs can reverse the inactivation of T cells, induced by T cell-specific immune checkpoints that silence them in the tumor microenvironment. However, the levels of checkpoint inhibitor drugs achieved in tumors are often not high enough to rekindle anti-tumor immunity, and clinicians fear that checkpoint inhibitor drugs could have unspecific adverse effects in the body, especially when administered in combinations.
Smith will investigate in mouse models whether tumor-specific CAR-T cells can be turned into living “micropharmacies” that produce and release checkpoint inhibitors locally in tumor microenvironments in order to enhance their effects in solid tumors and keep the level of drugs circulating in the body at a minimum.
“The i3 Center is excited to have this opportunity to add Dr. Smith to our team, as his scientific expertise and clinical perspective will provide a significant boost to our efforts to develop improved immunotherapies,” said Mooney, who leads the Wyss Institute’s Immuno-Materials Initiative, and also is the Robert P. Pinkas Family Professor of Bioengineering at the SEAS.
The i3 Center was founded by the Wyss Institute and its collaborating institutions, including SEAS, the Dana-Farber Cancer Institute, and Harvard’s Department of Stem Cell and Regenerative Biology as a cross-institutional and cross-disciplinary initiative in which world-leading researchers in the cancer immunology and bioengineering fields work together to create biomaterials-based approaches for more effective anti-cancer immunotherapy in settings where it currently is limited, such as in myeloid malignancies and solid tumors.
For more information about the Harvard i3 Center: Biomaterials to Create T Cell Immunity, please contact Eileen Barrette.
PRESS CONTACT
Wyss Institute for Biologically Inspired Engineering at Harvard University
Benjamin Boettner, benjamin.boettner@wyss.harvard.edu, +1 617-432-8232
Immuno-engineering to Improve Immunotherapy (i3) Center
Eileen Barrette, mailto:eileen.barrette@wyss.harvad.edu

